Tris(dibenzylideneacetone)dipalladium(0) CAS 51364-51-3
High-Purity Tris(dibenzylideneacetone)dipalladium(0) from Aure Chemical – Your Trusted Source
Tris(dibenzylideneacetone)dipalladium(0), Pd₂(dba)₃, CAS No. 51364-51-3, is one of the most widely used Pd(0) precatalysts in modern organometallic chemistry and synthetic organic synthesis. Supplied by Aure Chemical as a dark purple to black crystalline powder with high palladium content (approximately 21–22% Pd), this air-sensitive complex readily dissociates in solution to generate highly active Pd(0) species, especially in the presence of ligands. Renowned for its exceptional activity, broad functional group tolerance, and mild reaction conditions, it is the benchmark precursor for Suzuki-Miyaura, Heck, Sonogashira, Negishi, Buchwald-Hartwig amination/etherification, and various C–H activation reactions essential to pharmaceutical API synthesis, agrochemicals, and advanced materials. Aure Chemical provides rigorous purity control, low residual dba, and secure palladium sourcing to ensure batch consistency and optimal catalytic performance in research and industrial-scale processes worldwide.
Basic Information of Tris(dibenzylideneacetone)dipalladium(0)
Aure Chemical supplies Tris(dibenzylideneacetone)dipalladium(0) as a premium Pd(0) precatalyst with verified palladium content and exceptional purity for advanced cross-coupling and activation applications.
| CAS No. | 51364-51-3 |
|---|---|
| EC No. | 257-098-6 |
| Chemical Formula | Pd₂(C₁₇H₁₄O)₃ |
| Molecular Weight | 915.73 g/mol |
| Appearance | Dark purple to black crystalline powder |
| Odor | Odorless to slight characteristic |
| Melting point | Decomposes >150 °C |
| Boiling point | Not applicable (decomposes) |
| Density | approx. 1.4–1.6 g/cm³ |
| Solubility | Insoluble in water; soluble in toluene, THF, dichloromethane, chloroform (air-sensitive solutions) |
| Nature (hazards) | Harmful if swallowed, may cause long lasting harmful effects to aquatic life, air-sensitive |
| RIDADR | Not regulated (not subject to transport regulations as hazardous material in many classifications) |
| Chemical Structure | 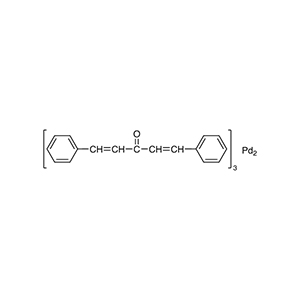 |
Aure Chemical provides customizable options including purity grades (99%+ Pd basis), dba content control, air-sensitive packaging under inert atmosphere, and specific batch sizes to preserve activity for your synthesis or scale-up needs.
Primary Applications of Tris(dibenzylideneacetone)dipalladium(0)
Tris(dibenzylideneacetone)dipalladium(0), commonly abbreviated as Pd₂(dba)₃, is a benchmark Pd(0) precatalyst delivering superior activity in ligand-assisted cross-coupling and functionalization reactions across industries. The weakly coordinating dibenzylideneacetone (dba) ligands are readily displaced under reaction conditions, generating highly reactive Pd(0) species suitable for systems described in palladium cross-coupling catalysis, where rapid oxidative addition and controlled catalyst activation are essential.
Suzuki-Miyaura & Other Cross-Couplings
Enables high-yield aryl-aryl, aryl-alkenyl, and aryl-alkynyl bond formation with broad substrate scope, serving as the gold standard in pharmaceutical and agrochemical intermediate synthesis. The preformed Pd(0) center accelerates oxidative addition into aryl halides, enhancing catalytic turnover under mild conditions.Buchwald-Hartwig Amination & Etherification
Facilitates efficient C–N and C–O bond construction under mild conditions, providing access to nitrogen- and oxygen-linked pharmacophores with excellent functional group compatibility. When combined with tailored phosphine ligands, Pd₂(dba)₃ forms highly active homogeneous catalytic systems.C–H Activation & Functionalization
Supports direct C–H arylation, olefination, and amidation, offering atom-economical routes to complex molecules in fine chemicals and drug discovery. Its well-defined Pd(0) state enables efficient coordination to directing groups and subsequent bond activation pathways.Heck & Carbonylative Couplings
Catalyzes vinylation of aryl halides and carbonylative processes to form ketones, esters, and amides, critical for specialty chemical and heterocyclic synthesis. The labile dba ligands facilitate rapid ligand exchange and active species formation.Pharmaceutical API Manufacturing
Widely applied in multi-kilogram-scale Pd-catalyzed steps of active pharmaceutical ingredients, delivering selectivity, scalability, and process robustness. Its predictable activation profile supports reproducible catalyst performance in regulated manufacturing environments.Catalyst Development & Mechanistic Studies
Serves as a standard Pd(0) source for ligand screening, mechanistic investigations, and development of novel palladium catalytic systems in R&D laboratories. The defined molecular structure allows systematic evaluation of ligand effects and catalytic pathways.
For a broader overview of palladium material categories—including salts, oxides, supported catalysts, and organometallic systems across industrial sectors—refer to Palladium Compounds: Applications, Categories & Industrial Uses.
Why Choose Aure Chemical for Your Tris(dibenzylideneacetone)dipalladium(0) Supply?
Partner with Aure Chemical for Tris(dibenzylideneacetone)dipalladium(0) to benefit from superior Pd(0) activity, technical expertise, and dependable palladium supply for your catalytic transformations.
High Purity & Air-Sensitive Handling
Advanced manufacturing and packaging under inert conditions preserve compound integrity, minimizing dba dissociation and ensuring maximum catalytic potency.Custom Precursor Specifications
Tailored Pd content, dba stoichiometry, purity levels, and packaging to perfectly align with your ligand system, solvent, and reaction scale requirements.Secure Palladium Supply Chain
Transparent, responsible procurement with inventory buffering to deliver supply stability and competitive pricing in dynamic palladium markets.Technical Catalysis Support
Dedicated specialists provide reaction optimization, ligand synergy advice, scale-up guidance, and troubleshooting to maximize yield and selectivity.Regulatory & Sustainability Compliance
Comprehensive COA, REACH, RoHS, SDS documentation and palladium recycling programs aligned with global standards and ESG commitments.
Hazards Classification
GHS Classification: Acute Toxicity, Oral (Category 4); Aquatic Chronic (Category 4)
Hazard Statements: H302: Harmful if swallowed; H413: May cause long lasting harmful effects to aquatic life
UN Number: Not regulated
Hazard Class: Not classified as hazardous for transport
Packing Group: Not applicable


Packaging, Logistics & Compliance Capabilities
Aure Chemical provides flexible packaging solutions, compliant logistics arrangements, and controlled storage practices to support laboratory-scale, pilot-scale, and commercial-scale global supply.
Packaging Options
- 25 kg / 50 kg bags (fiber drum, aluminum bag, HDPE drum)
- 100 kg / 200 kg steel or HDPE drums
- 500 kg / 1000 kg IBC containers
- Bulk liquids in ISO tank containers
- Customized packaging upon request
Transportation Modes
- Sea freight (FCL / LCL)
- Air freight for selected products
- Road and rail transportation
- Dangerous & non-dangerous cargo handling
- Incoterms: EXW, FOB, CFR, CIF, DAP, DDP
Storage & Handling
- Dry, cool, and well-ventilated warehouses
- Segregation by hazard class
- FIFO inventory management
- Temperature-controlled storage if required
- Regular inspection & labeling control
Documentation & Compliance
- Safety Data Sheet (SDS)
- Certificate of Analysis (COA)
- Dangerous Goods Declaration (DGD)
- Packing List & Commercial Invoice
- IMDG / IATA / ADR compliant labeling
For packaging customization or logistics consultation, please contact us.