
Sodium tetrachloropalladate(II) CAS 13820-53-6
High-Purity Sodium Tetrachloropalladate(II) from Aure Chemical – Your Trusted Source
Sodium tetrachloropalladate(II), Na₂[PdCl₄], CAS No. 13820-53-6, is a highly soluble anionic palladium(II) complex widely used as a convenient water-soluble precursor in palladium chemistry. Supplied by Aure Chemical as reddish-brown to dark brown crystalline powder or hydrated solid with high palladium content (approximately 36–37% Pd), this air-stable salt readily dissociates in aqueous or polar media to provide [PdCl₄]²⁻ ions. It serves as an efficient starting material for preparing palladium precatalysts employed in cross-coupling reactions (Suzuki, Heck, Sonogashira), C–H activation, allylic substitution, and palladium nanoparticle synthesis, as well as for electroless palladium plating and organometallic complex formation. Its excellent solubility and controlled reactivity make it preferred over anhydrous PdCl₂ in aqueous-phase processes and scale-up applications in pharmaceuticals, fine chemicals, and electronics. Aure Chemical ensures precise Pd assay, low impurity levels, and secure palladium sourcing to deliver reproducible performance for industrial and research use worldwide.
Basic Information of Sodium Tetrachloropalladate(II)
Aure Chemical provides Sodium Tetrachloropalladate(II) as a premium-grade anionic Pd(II) precursor with verified palladium content and exceptional purity for catalytic and plating applications.
| CAS No. | 13820-53-6 |
|---|---|
| EC No. | 237-502-6 |
| Chemical Formula | Na2PdCl4 |
| Molecular Weight | 294.21 g/mol (anhydrous basis) |
| Appearance | Reddish-brown to dark brown crystalline powder |
| Odor | Odorless |
| Melting point | Decomposes before melting (>300 °C) |
| Boiling point | Not applicable (decomposes) |
| Density | approx. 2.7–2.9 g/cm³ |
| Solubility | Highly soluble in water (>500 g/L); soluble in methanol, ethanol, acetone |
| Nature (hazards) | Causes severe skin burns and eye damage, may cause allergic skin reaction, harmful if swallowed, toxic to aquatic life |
| RIDADR | UN 3288, Toxic solid, inorganic, n.o.s. (Sodium tetrachloropalladate(II)), Class 6.1, Packing Group III |
| Chemical Structure | 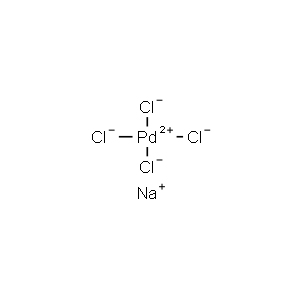 |
Aure Chemical offers customizable options including purity grades (99%+ Pd basis), hydration level control, and specialized packaging to meet your catalyst preparation, plating, or synthesis requirements.
Primary Applications of Sodium Tetrachloropalladate(II)
Sodium Tetrachloropalladate(II) serves as a water-soluble Pd(II) source, enabling efficient catalyst generation and palladium incorporation in aqueous or polar-phase processes. As a coordination complex containing the [PdCl₄]²⁻ anion, it provides controlled palladium delivery in solution and is frequently utilized in systems associated with palladium salt and catalyst precursor preparation, particularly where solubility and halide coordination influence catalytic performance.
Cross-Coupling Catalyst Preparation
Acts as a convenient precursor for generating Pd(II) precatalysts used in Suzuki-Miyaura, Heck, Sonogashira, and Negishi couplings, particularly in aqueous or biphasic systems for pharmaceutical intermediates. Under reducing conditions, [PdCl₄]²⁻ is converted to catalytically active Pd(0) species, enabling efficient C–C bond formation in scalable reaction environments.Palladium Nanoparticle Synthesis
Facilitates controlled reduction to produce stable Pd nanoparticles, colloids, or supported clusters for electrocatalysis, sensors, and hydrogenation applications with tunable size and dispersion. Its defined chloride coordination allows predictable nucleation and growth kinetics during nanoparticle formation.Electroless Palladium Plating & Surface Finishing
Provides Pd(II) ions for autocatalytic deposition of palladium layers on electronics, connectors, and circuit boards, offering excellent adhesion and corrosion resistance. The aqueous solubility and controlled redox behavior support consistent bath formulation and metal deposition quality.Organometallic Complex Synthesis
Serves as starting material for preparing Pd(II) complexes with phosphines, amines, carbenes, or other ligands in homogeneous catalysis and materials research. Ligand substitution reactions displace chloride ligands to form well-defined catalytic intermediates suitable for method development.C–H Activation & Allylic Substitution
Used to form active Pd species for direct C–H functionalization and Tsuji-Trost allylic alkylation, providing efficient routes to complex molecules in fine chemicals. Chloride coordination can modulate catalyst speciation and influence reaction selectivity under specific conditions.Aqueous-Phase Catalysis & Green Chemistry
Enables palladium-catalyzed reactions in water or aqueous media, supporting environmentally friendlier processes in pharmaceutical and specialty chemical production. Water-compatible catalytic systems reduce organic solvent usage and improve process sustainability.
For a broader classification of palladium materials—including salts, oxides, supported catalysts, and organometallic systems across industrial sectors—refer to Palladium Compounds: Applications, Categories & Industrial Uses.
Why Choose Aure Chemical for Your Sodium Tetrachloropalladate(II) Supply?
Aure Chemical supplies Sodium Tetrachloropalladate(II) with consistent palladium assay, controlled impurity profile, and technical support for catalyst preparation, plating, and materials development applications.
High Palladium Content Consistency
Rigorous analytical controls ensure accurate Pd assay, minimal metallic and trace impurities, and batch-to-batch reproducibility for dependable catalytic or plating performance.Secure Palladium Sourcing
Responsible procurement practices and inventory planning support stable supply amid precious metal market fluctuations.Technical Application Guidance
Technical assistance is available for dissolution protocols, reduction strategies, catalyst activation, plating bath optimization, and nanoparticle synthesis parameters.Full Regulatory Compliance
Comprehensive documentation including COA, REACH, RoHS, and SDS supports integration into regulated manufacturing and international trade environments.Sustainability & Recycling Support
Palladium recovery and recycling programs help improve material efficiency and support circular economy objectives.
Hazards Classification
GHS Classification: Skin Corrosion/Irritation (Category 1B); Serious Eye Damage (Category 1); Skin Sensitization (Category 1); Acute Toxicity, Oral (Category 4); Aquatic Chronic (Category 2)
Hazard Statements: H314: Causes severe skin burns and eye damage; H317: May cause an allergic skin reaction; H302: Harmful if swallowed; H411: Toxic to aquatic life with long lasting effects
UN Number: UN 3288
Hazard Class: 6.1 (Toxic substances)
Packing Group: III




Packaging, Logistics & Compliance Capabilities
Aure Chemical provides flexible packaging solutions, compliant logistics arrangements, and controlled storage practices to support laboratory-scale, pilot-scale, and commercial-scale global supply.
Packaging Options
- 25 kg / 50 kg bags (fiber drum, aluminum bag, HDPE drum)
- 100 kg / 200 kg steel or HDPE drums
- 500 kg / 1000 kg IBC containers
- Bulk liquids in ISO tank containers
- Customized packaging upon request
Transportation Modes
- Sea freight (FCL / LCL)
- Air freight for selected products
- Road and rail transportation
- Dangerous & non-dangerous cargo handling
- Incoterms: EXW, FOB, CFR, CIF, DAP, DDP
Storage & Handling
- Dry, cool, and well-ventilated warehouses
- Segregation by hazard class
- FIFO inventory management
- Temperature-controlled storage if required
- Regular inspection & labeling control
Documentation & Compliance
- Safety Data Sheet (SDS)
- Certificate of Analysis (COA)
- Dangerous Goods Declaration (DGD)
- Packing List & Commercial Invoice
- IMDG / IATA / ADR compliant labeling
For packaging customization or logistics consultation, please contact us.








