High-Purity 6-Acetoxy-7-Methoxy-3,4-Dihydroquinazolin-4(3H)-One CAS 179688-53-0 from Aure Chemical
6-Acetoxy-7-methoxy-3,4-dihydroquinazolin-4(3H)-one is a substituted quinazolinone derivative commonly utilized as a protected intermediate in heterocyclic synthesis. Its acetoxy group at the 6-position allows for selective functionalization in multi-step processes.
Primarily employed in controlled pharmaceutical development environments, this compound supports precise construction of quinazoline-based structures. Aure Chemical maintains reliable specifications to meet requirements for intermediate-grade materials.
Basic Information
| CAS No. | 179688-53-0 |
|---|
| EC No. | 808-050-2 |
|---|
| Linear Formula | C11H10N2O4 |
|---|
| Molecular Weight | 234.21 |
|---|
| Appearance | Very pale yellow to pale yellow crystal - powder |
|---|
| Melting Point | 297 °C (lit.) |
|---|
| Boiling point | 390.5±52.0 °C (Predicted) |
|---|
| Density | 1.39 g/cm³ (Predicted) |
|---|
| Solubility | Slightly soluble in hot DMF; limited in common solvents (typical reference) |
|---|
| Storage Conditions | Store in a cool, dark place, sealed, recommended <15°C |
|---|
| Chemical Structure | 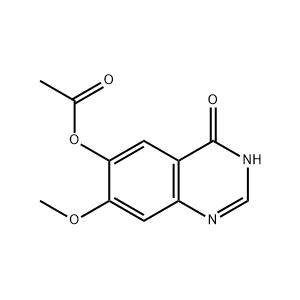 |
|---|
Key Applications
Gefitinib intermediate: Serves as a critical building block in the multi-step synthesis of gefitinib, an EGFR tyrosine kinase inhibitor used in oncology, and fits naturally within Gefitinib, Sunitinib and Sorafenib intermediates for targeted therapy development.
Pharmaceutical synthesis: Employed in the preparation of quinazoline-based active pharmaceutical ingredients and related therapeutic compounds, especially among oncology drug intermediates used in anticancer API synthesis.
Heterocyclic construction: Provides a protected quinazolinone scaffold for further elaboration in aromatic and nitrogen-containing molecule assembly, making it relevant to quinazolinone, benzimidazole and fused heterocycle intermediates.
Process development: Utilized in laboratory route scouting, optimization, and scale-up studies for fine chemicals and pharmaceutical intermediates for API synthesis.
Custom synthesis: Applied in bespoke projects requiring high-purity acetoxy-substituted quinazolinones for research and advanced pharmaceutical development.
Aure Chemical: Your Reliable Partner
Product quality consistency: Batch-to-batch uniformity maintained through rigorous in-house quality controls suitable for pharmaceutical intermediate applications.
Global supply stability: Dependable export capabilities with established channels ensuring timely delivery across scales from lab to pilot quantities.
Documentation support: Comprehensive SDS, COA, and related certificates readily provided to facilitate sourcing, compliance, and quality verification.
Technical and packaging service: Expert assistance available for technical inquiries, along with flexible options such as 1kg/5kg/25kg fiber drums with PE liners or customized packaging.

